Designing an optical system means knowing exactly how wavelength, frequency, and photon energy relate — and getting any one wrong cascades into every downstream component selection. Use this Frequency of Light Interactive Calculator to calculate frequency, wavelength, and photon energy using wavelength, frequency, or energy as your starting input, across any propagation medium. It's essential for optical communications, spectroscopy, and laser system design where a 0.01 nm wavelength error can push a DWDM channel off-grid. This page includes the governing formulas, a worked DWDM engineering example, full theory on wave-particle duality and dispersion, and a detailed FAQ.
What is the frequency of light?
The frequency of light is how many times per second an electromagnetic wave oscillates. Higher frequency means more energy per photon and a shorter wavelength — visible light sits in the range of roughly 430 to 770 trillion cycles per second (THz).
Simple Explanation
Think of light as a wave on water — frequency is how many wave crests pass a fixed point every second, and wavelength is the distance between crests. Speed is fixed in a given material, so if frequency goes up, wavelength must come down. Photon energy is just a measure of how much punch each individual packet of light carries, and it scales directly with frequency.
📐 Browse all 1000+ Interactive Calculators
Contents
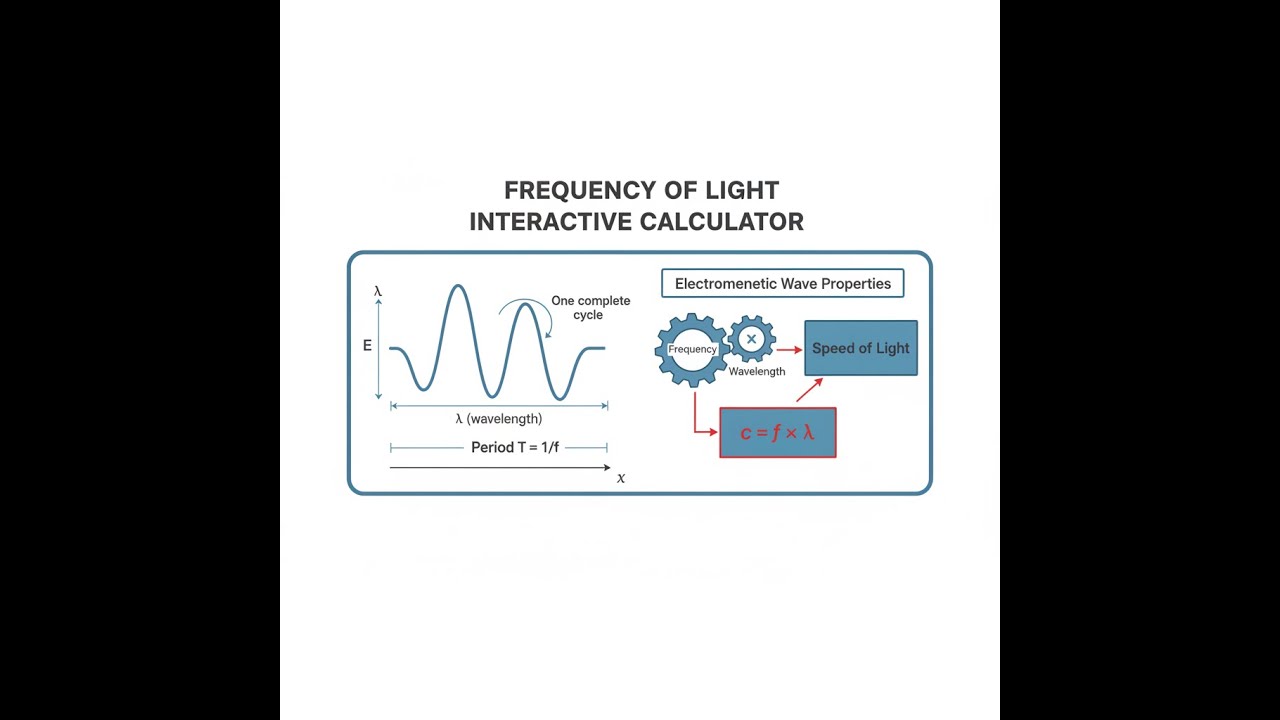
Electromagnetic Wave Diagram

How to Use This Calculator
- Select your calculation mode from the dropdown — choose what you want to solve for and what you already know.
- Enter your known value (wavelength in metres, frequency in Hz, or photon energy in J or eV) in the input field that appears.
- Select the propagation medium (vacuum, water, glass, diamond, or enter a custom refractive index).
- Click Calculate to see your result.
Frequency of Light Calculator
Frequency of Light Interactive Visualizer
Visualize how wavelength, frequency, and photon energy relate in electromagnetic waves. Watch the wave propagation change as you adjust parameters, and see how different media affect wavelength while frequency remains constant.
FREQUENCY
545 THz
ENERGY
2.25 eV
IN MEDIUM
550 nm
FIRGELLI Automations — Interactive Engineering Calculators
Governing Equations
Use the formula below to calculate frequency, wavelength, or photon energy from any starting electromagnetic wave parameter.
Fundamental Wave Equation
c = f × λ
Where:
- c = speed of light in vacuum = 2.998 × 108 m/s
- f = frequency of the electromagnetic wave (Hz)
- λ = wavelength in vacuum (m)
Photon Energy
E = h × f = (h × c) / λ
Where:
- E = photon energy (J or eV)
- h = Planck's constant = 6.626 × 10-34 J·s
- f = frequency (Hz)
- λ = wavelength (m)
Wave Propagation in Media
v = c / n = f × λmedium
λmedium = λvacuum / n
Where:
- v = wave speed in the medium (m/s)
- n = refractive index of the medium (dimensionless, n ≥ 1)
- λmedium = wavelength in the medium (m)
- λvacuum = wavelength in vacuum (m)
Note: Frequency remains constant across media boundaries; only wavelength and speed change.
Simple Example
Green light has a wavelength of 550 nm (5.5 × 10⁻⁷ m) in vacuum.
- Frequency: f = c / λ = 2.998 × 10⁸ / 5.5 × 10⁻⁷ = 5.45 × 10¹⁴ Hz (545 THz)
- Photon energy: E = hf = 6.626 × 10⁻³⁴ × 5.45 × 10¹⁴ = 3.61 × 10⁻¹⁹ J (2.25 eV)
- In glass (n = 1.52): wavelength shrinks to 550 / 1.52 = 362 nm; frequency stays at 545 THz.
Theory & Practical Applications
Electromagnetic Wave Nature and the Wave-Particle Duality
Light exhibits a fundamental dual nature that manifests differently depending on the experimental context. The wave model, described by Maxwell's equations, treats light as oscillating electric and magnetic fields perpendicular to each other and to the direction of propagation. The frequency of these oscillations defines the color perceived by the human eye for visible wavelengths (approximately 430-770 THz) and determines energy content for all electromagnetic radiation. The particle model treats light as discrete packets of energy called photons, each carrying energy E = hf where h is Planck's constant.
A critical distinction often overlooked in basic treatments: while frequency remains invariant when light crosses material boundaries, wavelength decreases by the refractive index ratio. This has profound implications for optical resonator design. A Fabry-Pérot cavity resonant at 1550 nm in vacuum will support standing waves at λ/n = 1019 nm when filled with silicon (n = 3.48 at telecom wavelengths). Engineers designing integrated photonic circuits must account for this wavelength compression when calculating resonator dimensions, waveguide coupling distances, and distributed Bragg reflector periods.
Frequency-Dependent Phenomena in Real Systems
Dispersion—the frequency dependence of refractive index—limits performance in high-bandwidth optical communication systems. Standard single-mode fiber (SMF-28) exhibits chromatic dispersion of approximately 17 ps/(nm·km) at 1550 nm. For a 100 Gbps signal with spectral width Δλ ≈ 0.8 nm transmitted over 80 km, pulse spreading reaches Δt = 17 × 0.8 × 80 = 1088 ps. Since the bit period is only 10 ps (1/100 Gbps), severe intersymbol interference occurs unless dispersion compensation is implemented.
The frequency-dependent absorption coefficient α(f) determines penetration depth in materials. For biological tissue, near-infrared wavelengths (700-900 nm, corresponding to 330-428 THz) penetrate several centimeters due to minimal absorption by hemoglobin and water. This "optical window" enables non-invasive deep-tissue imaging and photobiomodulation therapy. Conversely, ultraviolet-C radiation (200-280 nm, 1071-1500 THz) penetrates only micrometers into human skin but effectively disrupts viral and bacterial DNA—the physical basis for UV-C germicidal lamps.
Worked Engineering Example: Laser Diode Specification
A telecommunications engineer must specify a laser diode for a dense wavelength division multiplexing (DWDM) system operating on the ITU-T G.694.1 grid. The system requires a channel centered at 193.1 THz with maximum frequency drift of ±1.25 GHz under temperature variations from 0°C to 70°C.
Part A: Calculate the center wavelength in vacuum
Using c = f × λ and solving for λ:
λ₀ = c / f = (2.99792458 × 10⁸ m/s) / (193.1 × 10¹² Hz) = 1.55234 × 10⁻⁶ m = 1552.34 nm
This wavelength falls in the C-band (1530-1565 nm), commonly used for long-haul fiber optic transmission.
Part B: Determine the allowable wavelength drift
The frequency drift tolerance is Δf = ±1.25 GHz = ±1.25 × 10⁹ Hz. We must find the corresponding wavelength tolerance. Taking the differential of c = fλ:
c = fλ → 0 = f(dλ) + λ(df) → dλ/df = -λ/f
For small variations: Δλ ≈ -(λ/f) × Δf
Δλ = -(1.55234 × 10⁻⁶ m / 193.1 × 10¹² Hz) × (±1.25 × 10⁹ Hz) = ∓1.004 × 10⁻¹¹ m = ∓0.01004 nm
The allowable wavelength drift is ±0.010 nm (±10 pm). Note the negative sign indicates frequency and wavelength vary inversely.
Part C: Calculate photon energy and required output power
For quantum efficiency calculations, determine the photon energy:
E = hf = (6.62607015 × 10⁻³⁴ J·s) × (193.1 × 10¹² Hz) = 1.2793 × 10⁻¹⁹ J
Converting to electronvolts: E = (1.2793 × 10⁻¹⁹ J) / (1.602176634 × 10⁻¹⁹ J/eV) = 0.7985 eV
If the system requires 10 mW optical output power, calculate the photon emission rate:
Power = (number of photons/second) × (energy per photon)
Photon rate = P / E = (10 × 10⁻³ W) / (1.2793 × 10⁻¹⁹ J) = 7.82 × 10¹⁶ photons/second
This enormous photon flux (78.2 petaphotons/second) illustrates why classical wave descriptions suffice for most macroscopic optical systems—quantum discreteness becomes negligible when dealing with such large photon numbers.
Part D: Wavelength in fiber core
The fiber core has refractive index n = 1.4682 at 1552.34 nm. Calculate the wavelength inside the fiber:
λ_fiber = λ₀ / n = 1552.34 nm / 1.4682 = 1057.05 nm
This 32% wavelength reduction affects guided-wave phenomena such as modal cutoff wavelengths and effective mode field diameter. For a fiber with V-number calculated using vacuum wavelength, the actual guided mode experiences the compressed wavelength, affecting coupling efficiency to integrated photonic devices.
Applications Across Industries
Optical Communications: The 1550 nm wavelength (193.1 THz) serves as the primary carrier for global fiber optic networks due to minimal attenuation (0.2 dB/km) and compatibility with erbium-doped fiber amplifiers (EDFAs). System designers exploit the 1530-1565 nm C-band and 1565-1625 nm L-band to create 80+ DWDM channels spaced 50 GHz (0.4 nm) apart, achieving aggregate capacities exceeding 10 Tbps on a single fiber pair.
Medical Phototherapy: Light-tissue interactions are strongly frequency dependent. Blue light at 463 nm (648 THz) treats neonatal jaundice by photoisomerizing bilirubin. Red light at 660 nm (454 THz) and near-infrared at 850 nm (353 THz) penetrate 2-3 cm into tissue, stimulating mitochondrial cytochrome c oxidase and modulating cellular metabolism through photobiomodulation mechanisms still under investigation.
Semiconductor Manufacturing: Extreme ultraviolet lithography (EUVL) at 13.5 nm (22.2 PHz) enables <7 nm process nodes by reducing diffraction-limited feature sizes. The photon energy at this wavelength is 91.8 eV—sufficient to directly break chemical bonds in photoresists without requiring multi-photon processes. However, essentially all materials absorb strongly at this frequency, necessitating reflective optics with multilayer Mo/Si coatings and operation in high vacuum.
Spectroscopic Analysis: Atomic emission and absorption spectroscopy relies on characteristic frequencies corresponding to electronic transitions. The sodium D-line doublet at 589.0 nm and 589.6 nm (508.8 and 508.3 THz) arises from the 3²P₃/₂,₁/₂ → 3²S₁/₂ transitions. Frequency precision better than 1 MHz enables laser cooling of sodium atoms to microkelvin temperatures for quantum computing applications.
Radio Astronomy: The 21-cm hydrogen line at 1420.4 MHz arises from the hyperfine transition in neutral atomic hydrogen. This specific frequency allows astronomers to map galactic hydrogen distribution and measure cosmological redshifts. The corresponding photon energy (5.87 μeV) is far too small for chemical effects but perfectly suited for large-scale coherent detection with radio interferometry arrays.
Non-Ideal Behaviors and Engineering Limitations
Real electromagnetic sources exhibit finite spectral linewidth Δf due to fundamental and technical noise processes. For laser diodes, spontaneous emission contributes a Lorentzian linewidth component following the Schawlow-Townes formula, modified by the linewidth enhancement factor α. A typical 1550 nm distributed feedback (DFB) laser with 30 mW output has intrinsic linewidth ~100 kHz, but thermal fluctuations and injection current noise increase practical linewidth to 1-10 MHz unless active stabilization is employed.
The group velocity (dω/dk) differs from phase velocity (ω/k) in dispersive media, causing pulse envelope and carrier to propagate at different speeds. In optical fiber, second-order dispersion β₂ = d²k/dω² causes Gaussian pulses to broaden as √(1 + (z/L_D)²) where L_D = T₀²/|β₂| is the dispersion length and T₀ is the initial pulse width. For T₀ = 10 ps in standard fiber (β₂ ≈ -20 ps²/km at 1550 nm), dispersion length is merely 5 km—after which pulse broadening becomes significant.
For additional wave and optics calculations, explore the complete engineering calculator library.
Frequently Asked Questions
▼ Why does frequency remain constant while wavelength changes when light enters a different medium?
▼ How does the photon energy equation relate to the threshold current in laser diodes?
▼ What limits the accuracy of frequency measurements in real optical systems?
▼ Why do different spectral regions require fundamentally different detection technologies?
▼ How do dispersion effects become more severe at higher data rates even though frequency remains constant?
▼ What physical mechanism causes the refractive index to vary with frequency?
Free Engineering Calculators
Explore our complete library of free engineering and physics calculators.
Browse All Calculators →🔗 Explore More Free Engineering Calculators
About the Author
Robbie Dickson — Chief Engineer & Founder, FIRGELLI Automations
Robbie Dickson brings over two decades of engineering expertise to FIRGELLI Automations. With a distinguished career at Rolls-Royce, BMW, and Ford, he has deep expertise in mechanical systems, actuator technology, and precision engineering.
Need to implement these calculations?
Explore the precision-engineered motion control solutions used by top engineers.